Nevisense Indication for Keratinocyte Cancer
Since 2014, Nevisense has been indicated for melanoma detection, with over 250,000 examinations performed. It is also indicated for the detection of keratinocyte cancers, based on data from multiple clinical studies.
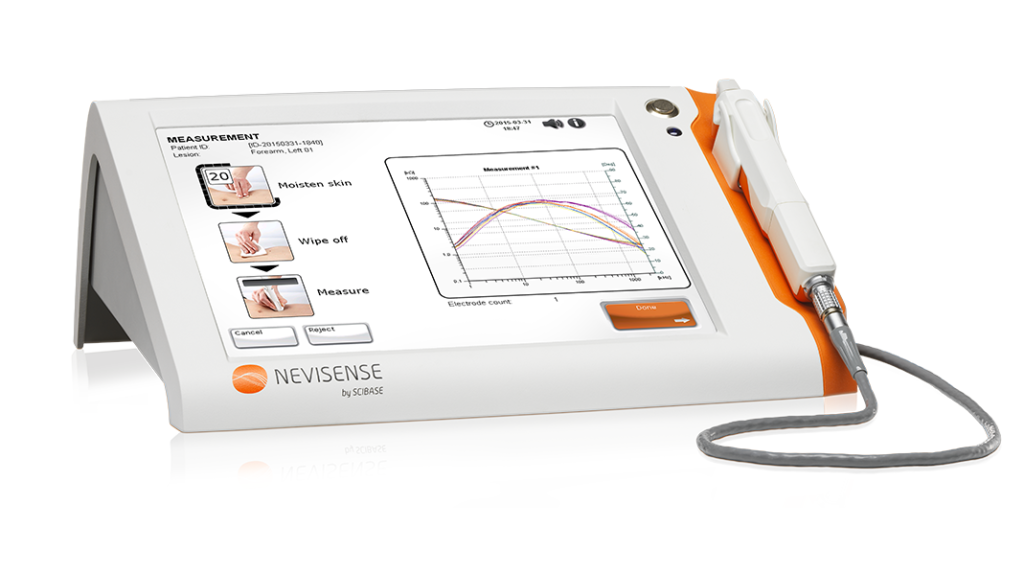
Measurement Procedure
In most cases, benign lesions and keratinocyte cancers can be confidently identified. For more complex cases, such as borderline or atypical lesions, additional information may be necessary. The Nevisense evaluation is used as part of a comprehensive risk assessment, alongside patient history and visual examination.
Nevisense provides an objective analysis, indicating the probability of malignancy based on extensive clinical study data. Physicians use this information in combination with a visual evaluation to make an informed clinical decision.

Nevisense Score for Keratinocyte Cancer
The Nevisense score is used to assess the risk of malignancy, with a positive score of 5 indicating an elevated risk. The Nevisense procedure is both safe and easy to use. To evaluate the lesion’s atypicality, measurements are taken after the skin is moistened with a saline solution.
Clinical Accuracy
- Three controlled clinical studies were conducted to assess the clinical performance of Nevisense in detecting keratinocyte cancers. These studies included:
• 123 cases of Basal Cell Carcinoma (BCC)
• 29 cases of Squamous Cell Carcinoma (SCC)
• 23 cases of Actinic Keratosis (AK)
• 1988 benign lesions
The results demonstrated the following clinical accuracy for keratinocyte cancers:
• Sensitivity: 100% [98.1% one-sided 95% confidence interval]
• Specificity: 53.8% [51.6%, 56.0% two-sided 95% confidence interval]
• Negative Predictive Value (NPV): 100%
• Positive Predictive Value (PPV): 14.7%

Boxplot of lesions types from all controlled studies
Boxplot of Lesion Types
In the boxplot shown, keratinocyte cancer (KC) lesions had an average EIS score of 8, melanoma had an average score of 7, and atypical lesions (including benign lesions and mild to moderate dysplastic nevi) had an average EIS score of 4.



